Marfan Syndrome: Decoding Aortic Risk from Fibrillin Mutation to Precision Medicine
Authors: Katharina Schatz, Ronny Schmidt, Monique Eutebach
Marfan Syndrome is a hereditary connective tissue disorder and a leading genetic cause of thoracic aortic aneurysm and dissection. Once considered primarily a structural disease, it is now recognised as a complex molecular signalling disorder driven by dysregulated TGF-β pathways, extracellular matrix remodelling and oxidative stress.
Explore how emerging biomarkers, together with high-throughput proteomics and antibody microarrays, are enabling molecular risk stratification and advancing Precision Medicine in Marfan Syndrome.
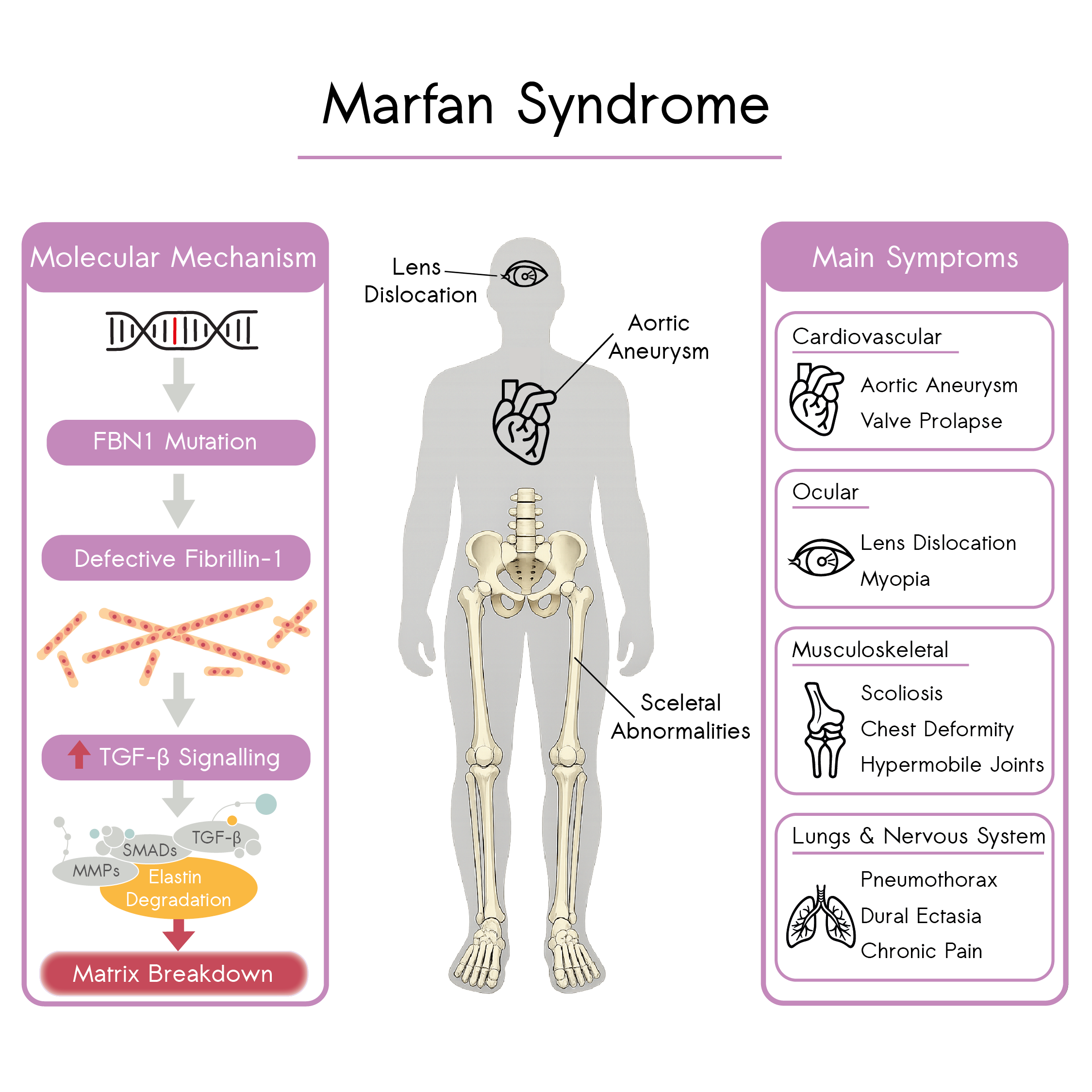
Molecular mechanisms and systemic manifestations of Marfan Syndrome: FBN1 mutations lead to defective fibrillin-1, dysregulated TGF-β signalling and extracellular matrix breakdown, driving aortic aneurysm formation and multisystem involvement affecting the cardiovascular, ocular, musculoskeletal and pulmonary systems.
Read more: Marfan Syndrome: Decoding Aortic Risk from Fibrillin Mutation to Precision Medicine
Stay tuned!

At Sciomics, we are dedicated to advancing healthcare through proteomics research.
We are collaborating with research groups and big pharma, alongside our in-house biomarker research initiatives.
To make this research more accessible to you, we launched the Sciomics science blog!
Register for our newsletter and follow us on LinkedIn to stay tuned!
Decoding Protein Complexity: The Fundamental Role of Post-Translational Modifications in Cellular Function and Disease
Authors: Henning Boekhoff, Jana S. Röder
Proteins are key players in cellular functions. They are encoded by their corresponding genes, but protein functions and activity are remarkably diverse and dynamic, largely due to post-translational modifications (PTMs). PTMs are covalent chemical changes to the side chains of amino acids that occur after protein synthesis, enhancing protein diversity across life forms from Archaea to Eukaryotes. This mechanism complements the complex network of proteomic regulation, greatly expanding upon gene expression, gene duplication, and alternative splicing. While the genome acts as a blueprint, providing the necessary information to build a functioning cell, the proteome is a complex structure that vastly extends beyond the blueprint’s scope.

Protein functions and activity are remarkably diverse and dynamic, largely due to post-translational modifications (PTMs). PTMs are covalent chemical changes to the side chains of amino acids that occur after protein synthesis, enhancing protein diversity across life forms from Archaea to Eukaryotes. This mechanism complements the complex network of proteomic regulation, greatly expanding upon gene expression, gene duplication, and alternative splicing.
Read more: Decoding Protein Complexity: The Fundamental Role of Post-Translational Modifications in Cellular...
The Parkinson’s Revolution: How Proteomics and Biomarkers Transform Diagnosis and Development of Disease-Modifying Therapies
Authors: Ronny Schmidt, Monique Eutebach
Parkinson’s Disease remains one of the most complex neurodegenerative disorders, driven by diverse molecular mechanisms and marked heterogeneity across patients. In this article, we explore how advances in proteomics and biomarker research are reshaping our understanding of this α-Synuclein pathology, improving diagnostic accuracy, and accelerating the development of disease-modifying therapies. Discover how Precision Medicine, molecular stratification, and innovative analytical technologies are paving the way toward earlier detection and truly targeted interventions in Parkinson’s Disease.

Key molecular and cellular hallmarks of Parkinson’s Disease reflect the multifactorial nature of neurodegeneration: α-Synuclein aggregation, neuroinflammation, mitochondrial dysfunction, oxidative stress, impaired proteostasis, and gut–brain axis involvement drive disease progression and define critical targets for biomarker discovery and precision therapy development.
Read more: The Parkinson’s Revolution: How Proteomics and Biomarkers Transform Diagnosis and Development of...
Biomarker Discovery for Predicting Dengue Deterioration: A Breakthrough Study
Authors: Jana S. Röder, Henning Boekhoff
Dengue is caused by the dengue virus (DENV), which is transmitted by multiple Aedes mosquito species. The disease has spread globally due to globalization, but infections are currently most common in tropical and subtropical regions. Endemic transmission of the dengue virus is mainly reported in the Eastern Mediterranean, the Americas, Southeast Asia, the Western Pacific, and Africa. However, transmissions have also been reported in Europe and the United States. In 2023, the World Health Organization (WHO) reported a near historic peak of dengue cases: there were over five million cases and more than 5000 dengue-related deaths. Close to 80 % of these cases, or 4.1 million, have been reported in the region of the Americas. The WHO states that the actual disease burden is likely higher, due to under-reporting of infections. It is estimated that more than half of the world’s population lives in areas affected by dengue virus. Dengue is a global threat.
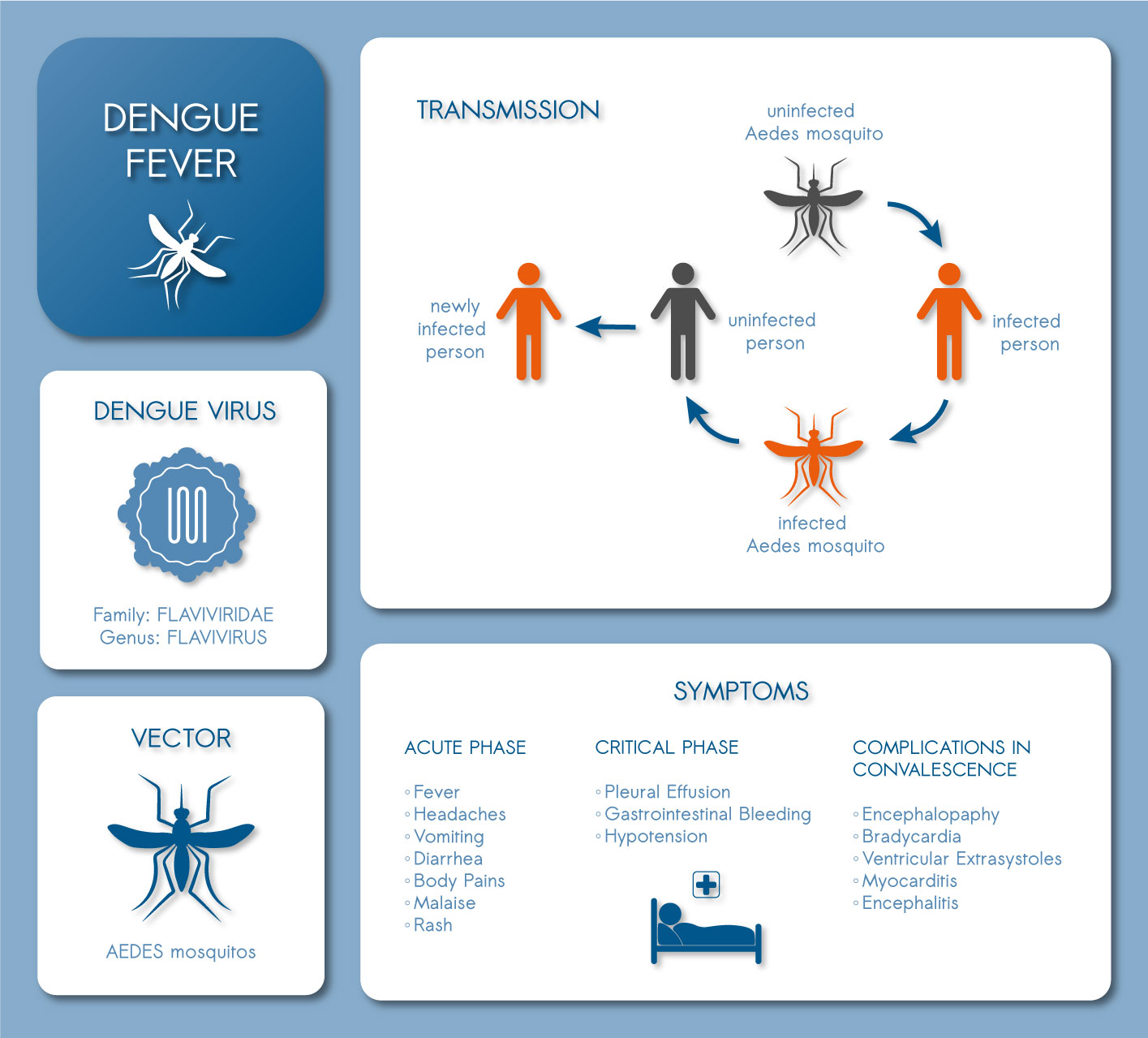
Dengue is a disease caused by dengue virus (DENV), which is transmitted by Aedes species. Dengue is a global threat. Simple and inexpensive strategies for the identification of patients with severe or life-threatening illness are urgently needed.
More Articles ...
Page 1 of 2
Testimonials
PD Dr. med. Mascha O. Fiedler-Kalenka and Dr. med. Benjamin Seybold
Heidelberg University Hospital, Department of Anesthesiology, Heidelberg, GermanyGerman Center for Lung Research (DZL), Translational Lung Research Center Heidelberg (TLRC), Heidelberg, Germany
"From the very beginning, working with the friendly and highly competent team at Sciomics was both professional and refreshingly straightforward. We particularly valued their openness to our specific research questions and the expert support they provided in planning and interpreting our exploratory cytokine analysis in a preclinical lung injury model. Communication was always on equal footing, enriched by Sciomics’ deep expertise, which also sparked new ideas for future projects."
Product: scioCD
Tara Sigdel, PhD
Associate Professor, Department of Surgery, University of California San Francisco
"We collaborated with Sciomics GmbH in Germany to analyze proteomics data from blood samples of kidney transplant recipients and liver perfusates during normothermic machine perfusion. Throughout the process, Sciomics provided exceptional support, ensuring timely data delivery and valuable assistance with data analysis. Their expertise and commitment have been instrumental in advancing our research, and we are currently using their data in multiple manuscript submissions. I highly recommend Sciomics for their reliable service and expertise in proteomics."
Product: scioDiscover