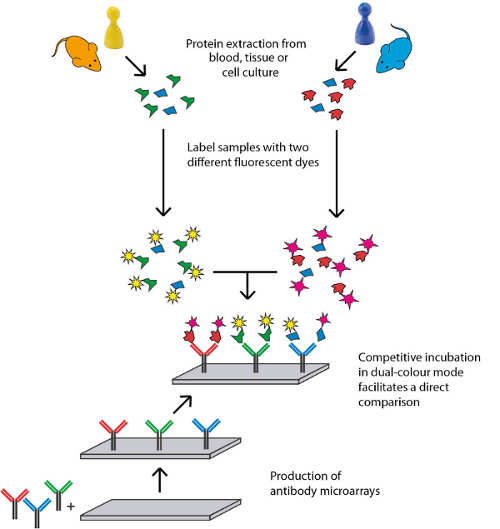
High quality standards are essential for robust and reliable Biomarker Discovery and Verification as well as for the discovery of new Drug Targets. During the last 10 years a stringent quality assurance workflow has been established for our Antibody Array Platform in order to conduct immuno-based biomedical discovery studies with excellent standards in terms of reproducibility and platform stability. The automated assay workflow further improves the performance of our service.
Biomarker Discovery Phase - scioDiscover Platform
- Stringent antigen design for maximised specificity of the resulting antibodies
- Antibodies are antigen affinity-purified
- Targets identified through transcriptional studies and expert input
- Stringent QC management
- Features:
- cross-species activity (human and mouse for high versatility)
- detection of several protein isoforms
- cost-efficiency through high content
Verification Phase - Custom Array Platform
- Stringent antigen design for maximised specificity of the resulting antibodies
- Antibodies are antigen affinity-purified
- In-house QC on Western Blot (different cell lines or tissues) and optional ELISA-based tests
- In-house QC on large-scale protein arrays (> 9.000 human proteins)
- In-depth validation of array performance
- Definition of biomarker signature with highest accuracy
- Selection of appropriate binders / sandwich pairs
- Features:
- specifically designed antibodies for further use in diagnostic assays
- fully renewable antibody formats
- in-depth quality controlled (WB, ELISA)
- comprehensive cross-reactivity profile (large-scale protein arrays)
Orthogonal Biomarker Validation
Verified Candidates can be further validated by orthogonal methods according to customer’s requirements
- Western Blotting ( WB )
- ELISA
- Immuno-Precipitation ( IP )
- Mass spectrometry ( MS )
- Biacore
Overview
Scio-Discover Biomarker
Discovery Custom Microarrays
for Biomarker Verification Contact
us